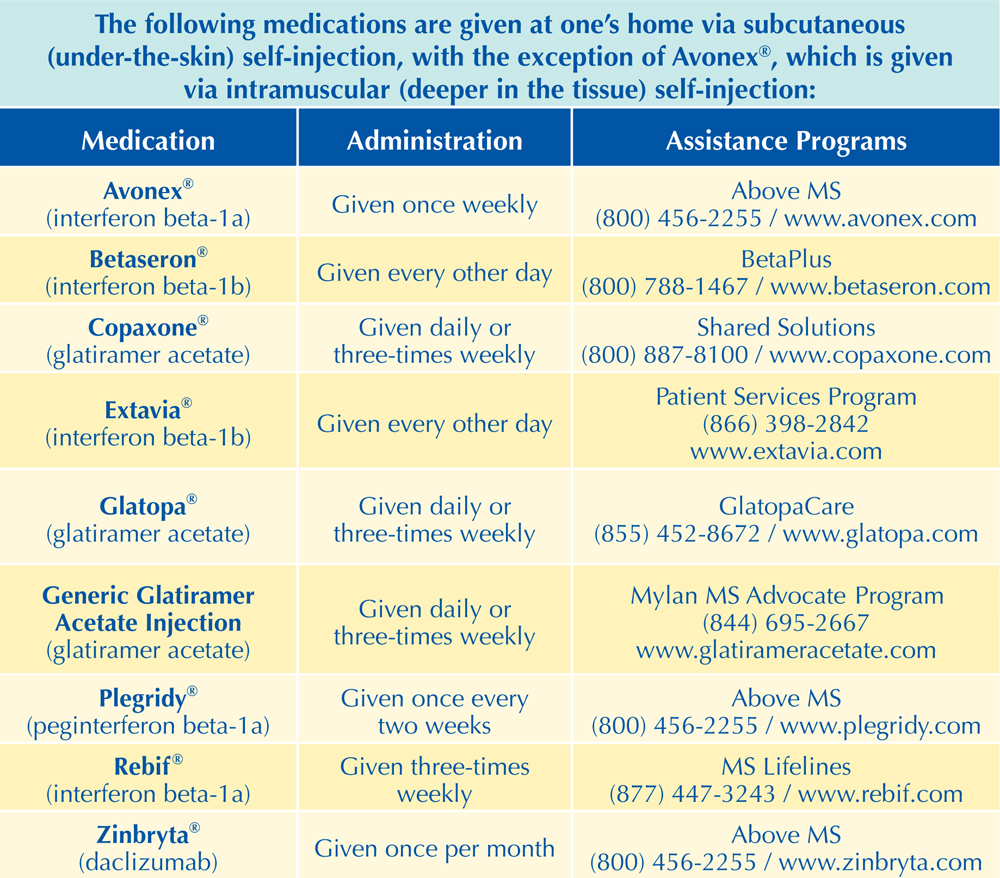
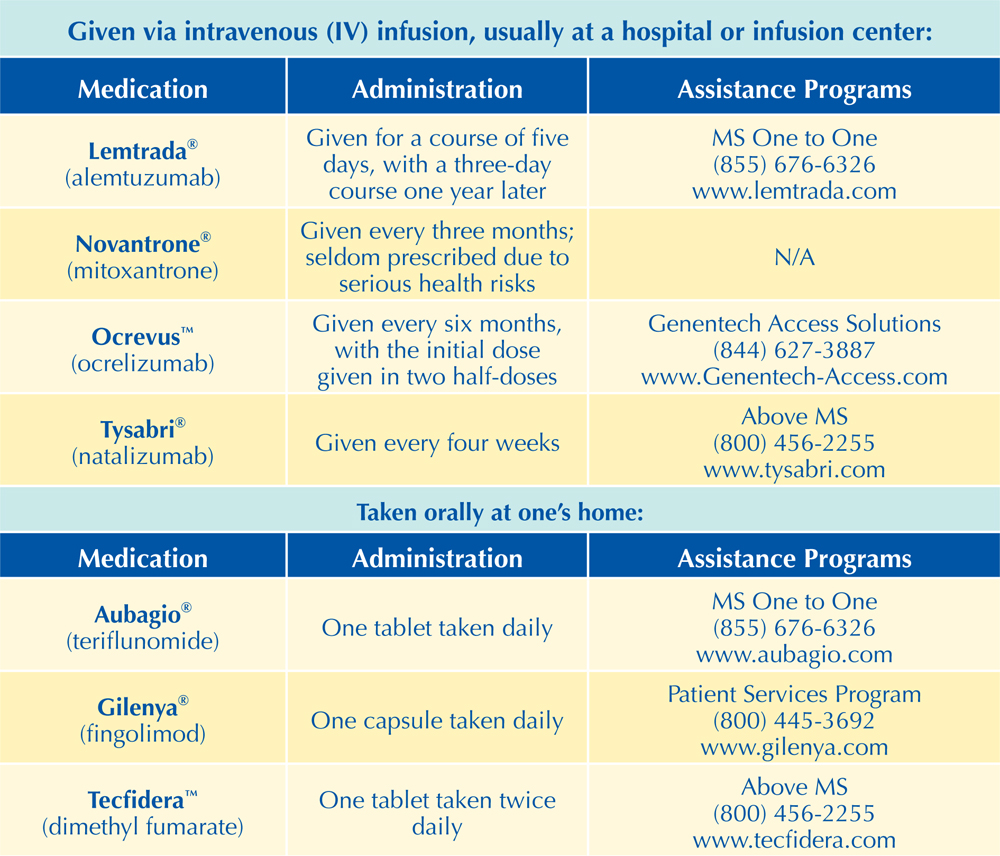
The FDA-Approved Long-Term Treatments for MS
The following chart includes the 16 disease-modifying therapies (DMTs) currently (at the time of printing) approved by the Food and Drug Administration (FDA) for the long-term treatment of MS. Additional DMTs are likely to be approved by the FDA in the near future. All of these DMTs are approved for treating either relapsing-remitting MS (RRMS) or all relapsing forms of MS, with the exception of Ocrevus™, which is also approved for primary-progressive MS (PPMS). Some have also been approved for “clinically isolated syndrome” (CIS), which refers to the initial symptom a patient reports prior to a diagnosis of MS.
As noted earlier, DMTs work to slow disease activity by reducing the number and severity of symptom flare-ups, reducing the number of active lesions that appear on an MRI, and potentially delaying the progression of the disease (possibly delaying any related disability). For more information on long-term treatments for MS, including any new approvals, please visit MSAA’s website at mymsaa.org to see the latest news, or call MSAA’s Helpline at (800) 532-7667, extension 154. Readers may also get information on how to select the most appropriate treatment through MSAA’s S.E.A.R.C.H.™ program at mymsaa.org/SEARCH.